The Stoffel lab's article on "The miR-200-Zeb1 axis regulates key aspects of B-cell function and survival in vivo " has just been published in Molecular Metabolism.
Highlights
- The double negative feedback loop of miR-200 and Zeb1 is essential for pancreatic B-cell function.
- Disruption of the miR-200-Zeb1 axis leads to broad transcriptional changes in islets affecting B-cell identity, EMT, insulin secretion and cell-cell junctions.
- Impaired inhibition of Zeb1 by miR-200 protects against ER stress.
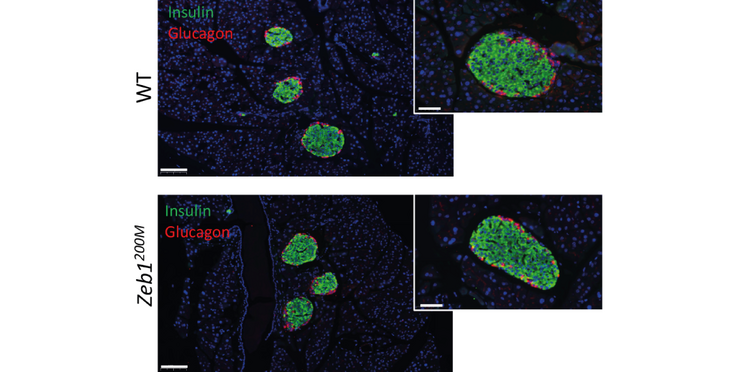
- Derepression of Zeb1 leads to impaired B-cell aggregation of dissociated islets and insulin secretion.
Abstract
Objective: The miR-200-Zeb1 axis regulates the epithelial-to-mesenchymal transition (EMT), differentiation, and resistance to apoptosis. A better understanding of these processes in diabetes is highly relevant as B-cell dedifferentiation and apoptosis contribute to loss of functional B-cell mass and diabetes progression, while EMT promotes loss of B-cell identity in in vitro expansion of human islets. While the miR-200 family has previously been identified as a regulator of B-cell apoptosis in vivo, studies focusing on Zeb1 are lacking. The aim of this study was thus to investigate the role of Zeb1 in B-cell function and survival in vivo.
Methods: miR-200 and Zeb1 are involved in a double-negative feedback loop. We characterized a mouse model in which miR-200 binding sites in the Zeb1 3'UTR are mutated (Zeb1200), leading to a physiologically relevant upregulation of Zeb1 mRNA expression. The role of Zeb1 was investigated in this model via metabolic tests and analysis of isolated islets. Further insights into the distinct contributions of the miR-200 and Zeb1 branches of the feedback loop were obtained by crossing the Zeb1200 allele into a background of miR-141?200c overexpression.
Results: Mild Zeb1 derepression in vivo led to broad transcriptional changes in islets affecting B-cell identity, EMT, insulin secretion, cell-cell junctions, the unfolded protein response (UPR), and the response to ER stress. Aggregation and insulin secretion of dissociated islets of mice homozygous for the Zeb1200 mutation (Zeb1200M) were impaired, and Zeb1200M islets were resistant to thapsigargin-induced ER stress ex vivo. Zeb1200M mice had increased circulating proinsulin levels but no overt metabolic phenotype, reflecting the strong compensatory ability of islets in maintaining glucose homeostasis.
Conclusions: This study points to the importance of the miR-200-Zeb1 axis in regulating key aspects of B-cell function and survival. A better understanding of this axis is highly relevant in developing therapeutic strategies for inducing B-cell redifferentiation, as well as maintaining B-cell identity in in vitro islet expansion.
Read the Publication in Molecular Metabolism (Open Access)
Abstract, figure and title from Title et al. (2021) Molecular Metabolism published under a CC BY-NC-ND 4.0 license.