The Mühlemann lab wrote a review on translational readthrough in eukaryotes. They discuss the factors that influence translational readthrough such as sequence context, tissue, age and others. They also summarize approaches to modulate readthrough for therapies by describing the mechanisms of different molecules and clinical trials involving them. Their review “Molecular determinants and therapeutic targeting of stop codon readthrough in eukaryotic translation ” has been published in the Journal of Molecular Biology.
Highlights
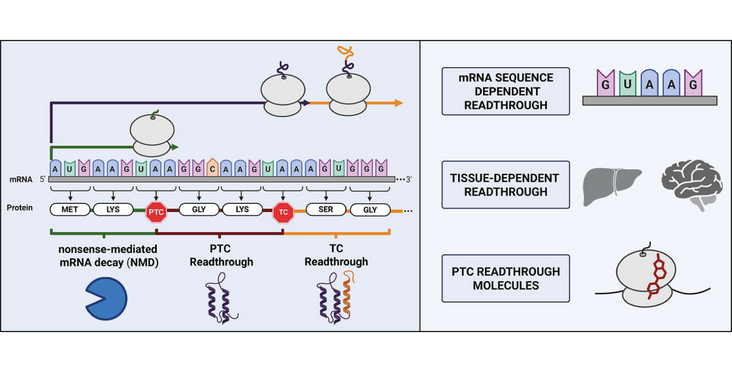
- Upstream and downstream nucleotide contexts shape stop-codon readthrough efficiency.
- Translational readthrough demonstrates marked tissue specificity and age dependence.
- PTC readthrough molecules act through distinct mechanisms.
Abstract
Accurate translation termination is essential for proteome integrity and is primarily governed by the release factors eRF1 and eRF3, which ensure precise recognition of stop codons and efficient release of nascent polypeptides. However, proteome integrity is challenged by mutations that generate premature termination codons (PTCs), leading to truncated, nonfunctional proteins and degradation of the aberrant transcript via nonsense-mediated mRNA decay (NMD). Collectively, these events account for ~1800 human genetic diseases.
Translational readthrough, the process by which near-cognate tRNAs decode stop codons and allow ribosomes to continue elongation beyond the stop codon, represents a possibility to suppress PTCs and restore full-length protein synthesis. Initially discovered in viruses as a mechanism to expand coding capacity, readthrough is now recognized as a regulated feature of eukaryotic gene expression influenced by both cis-acting sequence elements and trans-acting factors. Recent evidence highlights the remarkable context dependence of readthrough, revealing variation across transcripts, tissues, and developmental stages.
In this review, we examine the molecular determinants that define stop codon recognition and readthrough efficiency, with particular emphasis on nucleotide context. We further discuss the mechanisms and binding sites of small molecules that promote PTC readthrough, and summarize the clinical development landscape of readthrough-inducing compounds for the treatment of diseases caused by nonsense mutations.
Read the Review in the Journal of Molecular Biology (Open Access)
Abstract, figure, highlights and title from Teodorowicz and Mühlemann (2026) J Mol Biol published under a CC BY-NC 4.0 license.