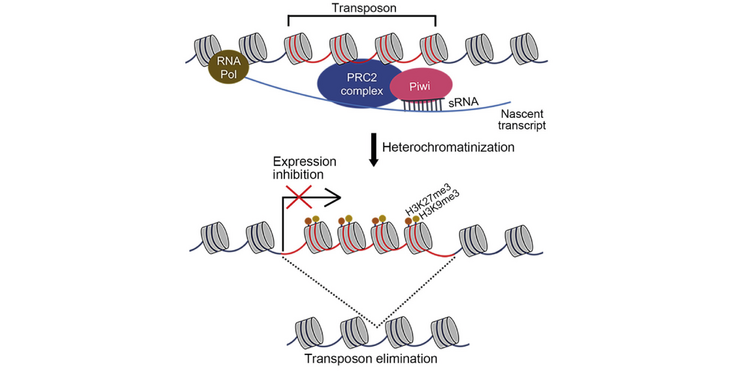
Genomes need to keep the transposons they contain silenced. In ciliates, the silencing occurs in the soma by excising transposons from the genomic DNA. The Nowacki lab (University of Bern) and collaborators found that the excision mechanism differs between old and new transposons. Namely, that only new transposons require heterochromation and small RNAs for excision. Their finding were published in the article "A small RNA-guided PRC2 complex eliminates DNA as an extreme form of transposon silencing" in Cell Reports.
Highlights
- PRC2 couples Piwi-bound small RNAs to transposon elimination in the new soma
- Elimination of new and ancient transposon sequences depends on PRC2
- Loss of PRC2 leads to retention and de-repression of transposons
- Only new transposons require heterochromatin and small RNAs for their removal
Abstract
In animal germlines, transposons are silenced at the transcriptional or post-transcriptional level to prevent deleterious expression. Ciliates employ a more direct approach by physically eliminating transposons from their soma, utilizing piRNAs to recognize transposons and imprecisely excise them. Ancient, mutated transposons often do not require piRNAs and are precisely eliminated. Here, we characterize the Polycomb Repressive Complex 2 (PRC2) in Paramecium and demonstrate its involvement in the removal of transposons and transposon-derived DNA. Our results reveal a striking difference between the elimination of new and ancient transposons at the chromatin level and show that the complex may be guided by Piwi-bound small RNAs (sRNAs). We propose that imprecise elimination in ciliates originates from an ancient transposon silencing mechanism, much like in plants and metazoans, through sRNAs, repressive methylation marks, and heterochromatin formation. However, it is taken a step further by eliminating DNA as an extreme form of transposon silencing.
Read the Publication in Cell Reports (Open Access)
Abstract, figure, highlights and title from Wang and Solberg (2022) Cell Reports published under a CC BY 4.0 license.