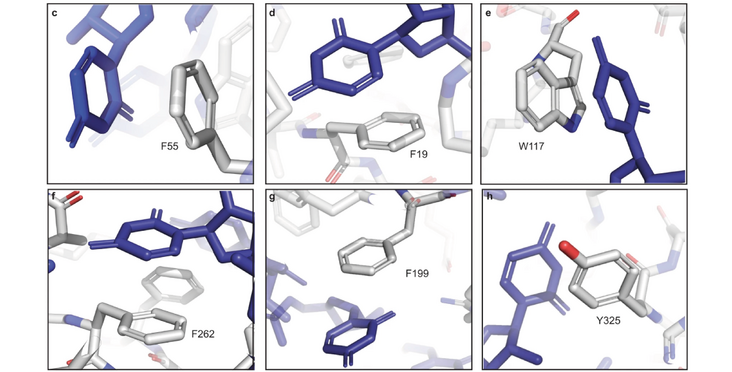
The three NCCR RNA & Disease platforms Structural MS (led by Alexander Leitner), NMR (led by Frédéric Allain) and RNA Synthesis (led by Jonathan Hall) joined forces to gain insights into the mechanim of protein RNA crosslinking. They discovered that RNA protein crosslinks require a Phenyalanine Pi-stacked to a nucleotide in their vincinity. Their findings should facilitate the analysis of cross-linking studies by Mass Spectrometry as well as CLIP datasets. The corresponding publication "Nucleotide-amino acid Pi-stacking interactions initiate photo cross-linking in RNA-protein complexes" has been published in Nature Communications.
Abstract
Photo-induced cross-linking is a mainstay technique to characterize RNA-protein interactions. However, UV-induced cross-linking between RNA and proteins at "zero-distance" is poorly understood. Here, we investigate cross-linking of the RBFOX alternative splicing factor with its hepta-ribonucleotide binding element as a model system. We examine the influence of nucleobase, nucleotide position and amino acid composition using CLIR-MS technology (crosslinking-of-isotope-labelled-RNA-and-tandem-mass-spectrometry), that locates cross-links on RNA and protein with site-specific resolution. Surprisingly, cross-linking occurs only at nucleotides that are Pi-stacked to phenylalanines. Notably, this Pi-stacking interaction is also necessary for the amino-acids flanking phenylalanines to partake in UV-cross-linking. We confirmed these observations in several published datasets where cross-linking sites could be mapped to a high resolution structure. We hypothesize that Pi-stacking to aromatic amino acids activates cross-linking in RNA-protein complexes, whereafter nucleotide and peptide radicals recombine. These findings will facilitate interpretation of cross-linking data from structural studies and from genome-wide datasets generated using CLIP (cross-linking-and-immunoprecipitation) methods.
Read the Publication in Nature Communications (Open Access)
Website Structural MS Platform
Abstract, figure and title from Knörlein et al. (2022) Nature Communications published under a CC BY 4.0 license.