The Ban and Schneider labs gained new insights into maturation of mitoribosomal small subunits in Trypanosomes. Their findings haven been published in the article "Mitoribosomal small subunit maturation involves formation of initiation-like complexes" in PNAS.
Significance
Mitochondria originated from ancestral bacteria and they still maintain their own reduced genome and translational apparatus that feature highly diverged mitoribosomes, specialized for the synthesis of membrane proteins. Only recently, high-resolution cryo-EM structures of the mature mitoribosomes from several species revealed their unusual structural features. Nevertheless, the mechanisms of their assembly are relatively poorly understood. To better understand how mitoribosomes are assembled and which factors participate in this process, we investigated assembly intermediates of the small mitoribosomal subunit from Trypanosoma brucei to reveal that the process involves a modular stepwise exchange of assembly factors that facilitate the formation of the functional decoding center of the small ribosomal subunit, and we discovered that mitochondrial initiation factor 2 participates in this process.
Abstract
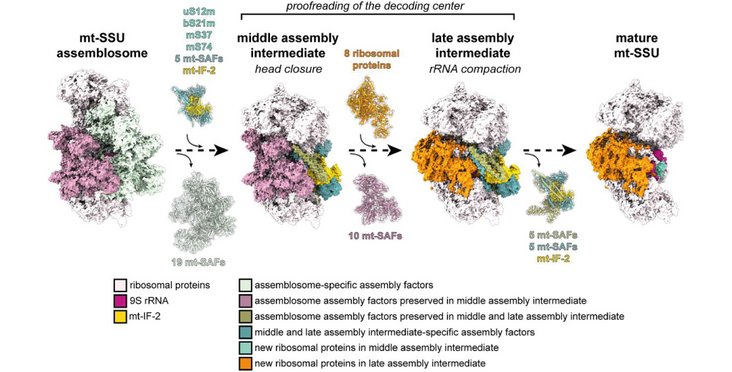
Mitochondrial ribosomes (mitoribosomes) play a central role in synthesizing mitochondrial inner membrane proteins responsible for oxidative phosphorylation. Although mitoribosomes from different organisms exhibit considerable structural variations, recent insights into mitoribosome assembly suggest that mitoribosome maturation follows common principles and involves a number of conserved assembly factors. To investigate the steps involved in the assembly of the mitoribosomal small subunit (mt-SSU) we determined the cryoelectron microscopy structures of middle and late assembly intermediates of the Trypanosoma brucei mitochondrial small subunit (mt-SSU) at 3.6- and 3.7-Å resolution, respectively. We identified five additional assembly factors that together with the mitochondrial initiation factor 2 (mt-IF-2) specifically interact with functionally important regions of the rRNA, including the decoding center, thereby preventing premature mRNA or large subunit binding. Structural comparison of assembly intermediates with mature mt-SSU combined with RNAi experiments suggests a noncanonical role of mt-IF-2 and a stepwise assembly process, where modular exchange of ribosomal proteins and assembly factors together with mt-IF-2 ensure proper 9S rRNA folding and protein maturation during the final steps of assembly.
Read the Publication in PNAS (Open Access)
Abstract, figure, synopsis and title from Lenarcic, Niemann et al (2022) PNAS published under a CC BY-NC-ND 4.0 license.