“You mean I am a liar or what?”

In this interview Steve Pascolo tells us about the history and perspective of mRNA therapeutics and his career.
A bit over 60 years ago, Yuri Gagarin was the first human in space. As far as you know, you were the first human to be injected synthetic mRNA coding for luciferase for experimental purposes: Do you see yourself as the Yuri Gagarin of mRNA therapies?
Not at all. He had to be much more courageous than me. RNA is quickly degraded, and similar experiments were conducted before in mice, so I knew there would be zero risk. The question was if we could detect luciferase activity. Besides, I was responsible of the pharmaceutic production of synthetic mRNA that I put in place in CureVac so I knew that the product is safe!
Nowadays, millions of humans are vaccinated with mRNA against SARS-CoV-2, but at the beginning there was a lot of skepticism as you indicate with the title of a recent review “Synthetic Messenger RNA-Based Vaccines: from Scorn to Hype”. Why that?
Because of a prejudice in the scientific and medical communities that had the textbook knowledge in their mind that mRNA is fragile. When I was showing the data to scientists, many of them were telling me “Ah no, it cannot work.” and I replied, “You mean I am a liar or what?” However, investors did not care about this prejudice when we could show them the data that mRNA-based vaccines work. A lot of people ignored the potential of mRNA vaccines even until as recent as 2019.
Did this scorn cause you a lot of frustration?
No, on the contrary. In a way, it was fun to fight. I knew that mRNAs will be one day used as drugs and will attract a lot of interest. From 1998 until like 2006, we as CureVac were alone, we were a small team and you are never so strong than when you are facing opposition. It creates a team spirit of taking up the challenge demonstrating them that it works. It was not frustration, on the contrary, it caused a type of energy.
The only thing that frustrated me came later relating to our early publications describing the luciferase injections into me and the first clinical study of mRNA vaccines (done in melanoma patients), which were published in 2007 and 2008 respectively. Both we only could publish in low impact factor journals. At the time, I did not care too much and was just happy that the results got published and the community had access to them. I got frustrated later, for example when our early work was not cited, although I know from personal communication that it inspired other vaccine developers in their experiments.
“…, we as CureVac were alone, we were a small team and you are never so strong than when you are facing opposition.”
When did you start to research mRNA vaccines?
For my postdoc, I went to the lab of Hans-Georg Rammensee in Tübingen. I started to compare peptide, protein and DNA based vaccines against cancer to see which one works best. In the Rammensee lab, I met Ingmar Hoerr, who was about to finish his PhD studies on mRNA vaccines. I went to Ingmar and told him that I would like to include mRNA vaccines in my comparison. Then, I tested the mRNA vaccine and it worked. However, it was less effective than the DNA one, but I was intrigued by the safety and production advantages it could provide.
Ingmar was not too much into science and wanted to do business. So, after his PhD he went to do an MBA but he was still then and when coming to the lab. During his visits, I would tell him about the results I had with mRNA vaccines. As part of his MBA, he had to write a business plan and he wrote it on an mRNA company. He then proposed to create CureVac. Ingmar and I were a good team, because I was really into science and he really into business while still understanding the science well.
What does it take to be a good entrepreneur?
I think you need to have a long-term big vision and self-confidence. You need to have this big vision that you will not build a house but a building. I am more the type that would build a studio, while Ingmar is more the type that wants to build a skyscraper with 150 floors.
“You need to have this big vision that you will not build a house but a building.”
You left CureVac in 2006 to go to the University Hospital Zurich (USZ) with a postdoc contract, have you ever regretted this decision?
No, not at all. I would have regretted leaving if later CureVac would have failed as a company. Then I would have thought “Oops, maybe if I would have stayed, the company would still exist.” When I left, I had the feeling that CureVac was on track and somebody else can take over as CSO. At that time, I did not like my job anymore because I spent all the time in the office and not doing any lab work. I was waking up and thinking “Damn, I have to go to work.” and that was a no go. So, I decided to go back to lab work, which is a lot of fun for me. In hindsight, I underestimated how much more data the regulatory authorities would still ask for and that the clinical studies still needed improvement. When I left, CureVac was the only company and only later competitors like BioNtech and Moderna emerged. In the end, I think it was not wrong to leave CureVac. The company did well without me, and I did more or less well without the company except the two years from 2010 to 2012 when I lost my job at the USZ.
Can you compare BioNtech, CureVac and Moderna?
The stories of BioNtech, CureVac and Moderna are quite different: At CureVac we were a team of basic scientists, who wanted to further develop and optimize mRNA vaccines bringing them into the clinic. The initial drive behind CureVac was scientific curiosity.
BioNtech had pure clinical roots: Özlem Türeci and Ugur Sahin were treating cancer patients. They founded two companies to help their patients because when they presented their ideas to pharmaceutical companies, the companies did not want to pursue these ideas.
In 2010, American investors felt that mRNA could do a lot, so they gave the capital and they hired people for developing the therapies. The Moderna history is business, which is good as well.
So, CureVac was scientific, BioNtech medical and Moderna I would say was business. I think it is good that these three companies have different energies driving their developments.
You state in interviews that you are not affiliated or have stocks neither in BioNtech, CureVac or Moderna. Still, you are listed as an inventor on 26 patents. Do you benefit from these financially?
Regarding the patents relating to CureVac, no. I gave up the rights of those to the company. The only patent I profit from is one that I filed as an USZ employee in 2008 on protamine-RNA nanoparticles, which was licensed and which I get a little bit of royalties from. I filed two more patents while being at the USZ. One on an optimized 5’UTR of the mRNA, which became obsolete after other researchers developed even better 5’ UTRs. In 2018, I filed a patent on a new type of chemically synthesized mRNA. If it gets approved and can be licensed, I could get royalties from. So, from the mRNA pioneers, I am probably the only one who will not get personally rich.
“I was waking up and thinking 'Damn, I have to go to work'.”
Do you expect that you will more easily acquire research funding?
Yes, that would be a great thing. The problem I have with my lab is financial sustainability and stability. I envisage at the University of Zurich (UZH) an mRNA department with three to four fixed positions so that we have a core of people whose salary is not dependent on successfully acquiring every two to three years the next grant. This would not only lead to stability but also institutional visibility, which is a goal I have so far not achieved.
If we get a few permanent positions and a real lab space for mRNA research, then we could plan projects long term. This would lead to patentable inventions, which could bring revenues for the University. This type of research is hard to do with the current unstable financial situation we have. With more stability we could really be part of the game and foster mRNA research in Zurich and Switzerland.
In April 2021, the only support I have from the UZH is from the University Research Priority Program Cancer (70'000 CHF per year till 2022), which started in 2016 and a small grant (28'200 CHF) for mRNA research in 2021. But there is no strong support from the USZ or UZH for mRNA research. I got an EU grant and an SNSF grant in 2020 on mRNA, which never happened in the past, but we now need to shift to second gear. We need a bigger lab and we need a GMP production facility for mRNA.
If Switzerland wants to have a BioNtech, CureVac or Moderna in the future, it is possible because there are big possibilities of using mRNA and great scientists in Switzerland but there needs to be support for this. Politicians still need to understand the big potential of mRNA and decide if Switzerland wants to be part of it. I talk to journalists so that the public and the politicians understand the mRNA technology in the vaccines but also beyond this, the great potential of the mRNA technologies in medicine.
Also, I suggested that the army should have an mRNA platform if they want to protect the Swiss population. Maybe the combat jets, which cost billions, are important but with a few millions invested into an mRNA platform including production, the Swiss people could be protected against emerging infectious diseases.
We need to see big and be part of the mRNA development in the world. This depends on political decisions to really go quick. And not only saying "We see the potential it has" and "Switzerland should be part of the development of mRNA therapies in the future."
“If Switzerland wants to have a BioNtech, CureVac or Moderna in the future it is possible.”
You developed in your lab also an mRNA vaccine against SARS-CoV-2 and injected it into mice. Did it work as far as you could tell? And if yes, were you tempted a year ago to vaccinate yourself with it?
Yes, the vaccine according to the animal experiments worked. And no, I was not tempted to vaccinate myself with it because the liposome we have is designed for vaccinating cancer patients by intravenous injection yielding mostly type 1 interferon and T cell response. It is not suitable if you want to have high levels of antibody titers. We lacked a good mRNA formulation for high level antibody production, but we are now working on that with the support by the Swiss National Science Foundation. Myself, I have recently received the second BioNtech shot, so in total I have received five injections of mRNA so far during my life.
What are the current research directions to optimize the mRNA technology?
One is to get the technology working with less and less mRNA required. The less you need to produce, the faster you can get the mRNA to billions of persons in need. Also, the distribution would be facilitated if formulations were found which allow to store the mRNA formulations for a long time at room temperature.
A lot of the pre-pandemic mRNA therapy research was focused on cancer therapies: Will we see a breakthrough in this area soon?
I hope so. BioNtech together with Genentech is currently conducting a Phase 2 trial with mRNA vaccines for treating melanoma. They combine the mRNA vaccine with an immune checkpoint inhibitor. So far, using a vaccine alone to treat a cancer has been shown to be not sufficient whether you use vaccines based on mRNA, peptides, proteins, viruses or DNA. So, it was not the mRNA itself that was the problem why the first mRNA vaccines against cancer did not work.
Nearly every vaccine type has been tested against cancer and none of them have been successful. They do induce an immune response but the immune response over time does not control the tumor. We need combinations and we will see how it evolves but the vaccines against cancer mutations developed by BioNtech are very potent because they on one hand give type I interferon response and on the other hand T cells directed against the tumor mutations. I hope this development will lead to approved mRNA vaccines against cancer. I think once the door is open there will be a lot of followers.
Also, mRNA has great potential for for Chimeric antigen receptor T cell therapies. The current CAR-T cell therapies are based on gene therapy with DNA. When you use mRNA, you have the advantage that it is transient, so you do not have the long-term risk of cells proliferating causing a cytokine storm or encephalitis as it has been seen in some patients.
When the tumor starts to escape by mutating and you want to redose the patient with DNA-modified T cells carrying a different CAR, you might have accumulation of the previously injected CAR-T cells increasing the risks of leukemia, cytokine storms and these types of adverse reactions. This concern does not exist with mRNA modified T-cells.
You can make with mRNA CAR-T cells that would be active for 2 or 3 days and dose the patient every week in order to get rid of the tumor. When the tumor becomes resistant to this CAR then you can continue treating with an mRNA coding for another CAR and continue the treatment without the risk of having accumulation of previously injected CAR T cells.
“You can jeopardize the development of a technology by not patenting it and just putting it into the public domain. ”
What other therapeutic areas do you see for mRNA-based therapies besides cancer and infectious diseases?
It is very broad. For example, mRNA is great for the area of regenerative medicine. If you want to regenerate damaged tissue, you want to do this temporarily. For this mRNA is perfect. On top of that, mRNA has great potential for personalized medicine and treating rare genetic diseases.
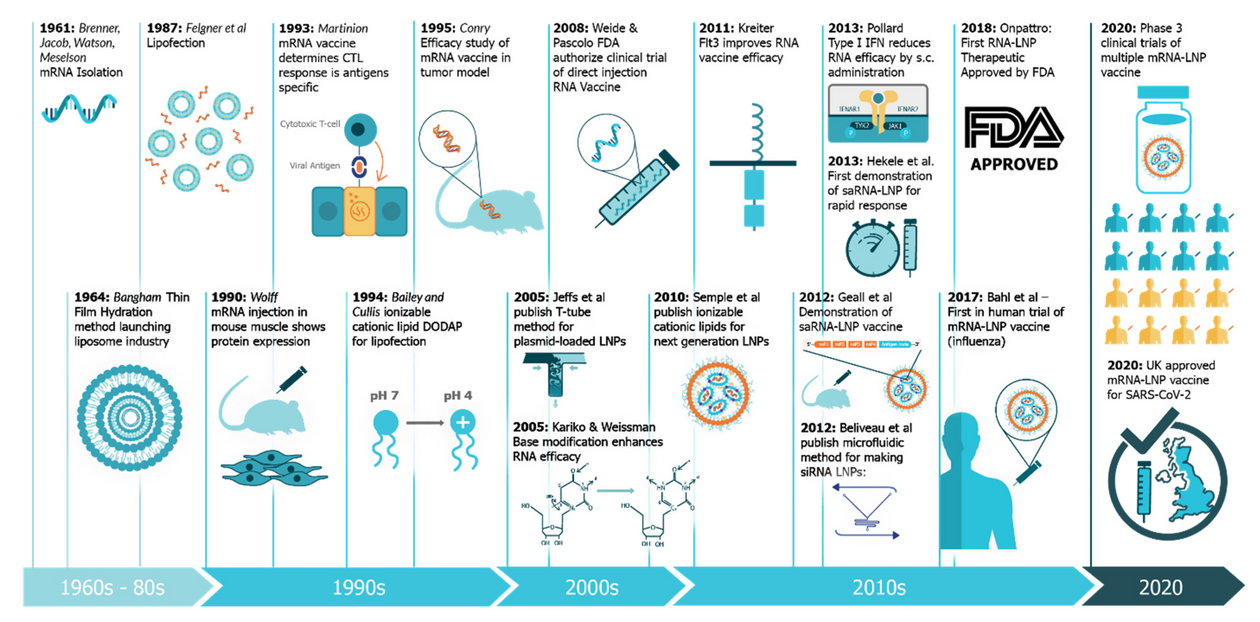
Back to the start of mRNA therapeutics research: In the media, Katalin Karikó and Drew Weissman are frequently mentioned as the scientists having made groundbreaking discoveries regarding mRNA vaccines. They pioneered the use of modified mRNA for therapy but not the use of mRNA as a vaccine?
Exactly, everybody talks about them, but this story is not complete like that. They disclosed in 2005 that modified mRNA is not immunostimulating, allowing it to be used for therapies - the exact opposite of vaccines: the injected mRNA does not trigger inflammation and consequently there is no immune response against the encoded protein. Kati has done a lot of great work on mRNA therapies, but to my knowledge, you do not find in the literature an article on mRNA vaccines with her as first or last author. Drew worked on mRNA vaccines and published in 2017 a Nature paper as last author on the usage of modified mRNA for vaccination. This came as a surprise, because modified mRNA was not supposed to cause inflammation, which is required for vaccination. So far it is not known whether modified or non-modified mRNAs are better for vaccines. The anti-cancer mRNA vaccines in development by BioNTech are all non-modified. I think this media attention comes from the fact that the communication of the Americans including their universities is unbeatable and we do not have this culture here.
I am always telling people that it started in 1990 by Wolff and colleagues showing for the first time that synthetic mRNA can be used in vivo to induce protein expression by injecting naked mRNA intramuscularly in mice. Regarding the use of mRNA for vaccination, the breakthrough was in 1993 when Martinon and colleagues published that mRNA produced in vitro and put into liposomes can be used as a vaccine.
However, this publication had also a bad side because the authors did not believe themselves in the technology to the extent that they filed a patent but then abandoned it. So, this led to using mRNA in liposomes as being in the public domain. When we founded CureVac we did not have the means to develop new patentable liposome formulations, so we started using protamines that we could get a patent on. You can jeopardize the development of a technology by not patenting it and just putting it into the public domain.
“That is horrifying to me because people were dying in vain.”
In the last couple of months, you were in my impression omnipresent in the media in France and Switzerland. Why are you doing this?
I see it as my role to inform the public in a neutral way. It is an advantage that I do not have shares from any of these companies and I can say freely what I think. We are now in a new dimension because of the problems with the AstraZeneca vector vaccine, and people want the mRNA vaccines. However, we should not forget that in December 2020, a lot of people were skeptical about mRNA vaccines. Then it was my role to explain to the general public what mRNA is and that it is degraded quickly, efficient and safe as vaccine, so that then everybody can decide themselves whether they want to get vaccinated with mRNA or not.
Also, I had to go to the media to counteract statements made by pseudo experts that were saying “Oh no, the mRNA vaccine: we do not know what is inside and we should wait”. That is horrifying to me because people were dying in vain. We had the vaccine and then “experts” come on TV seeding doubts in the public. There were some people on the radio saying that because of the skepticism seeded by the pseudo experts, they did not want their elderly father to be vaccinated, who could have been vaccinated with mRNA as early as January 2021. In March, he died from Covid-19. It was very important at that time to explain what mRNA is and stress that the whole development of mRNA vaccines and therapies is based on safety advantages of this format.
What were the reactions you got from the public after your media appearances?
Most reactions were positive and people wrote to thank me that they now understood what mRNA vaccines are, and that they will get vaccinated. This type of reactions showed to me that the time spent talking to and on the media was worth it. There was also a minority of persons accusing me that I do this media work for money, but I did not get any financial benefit from media or from mRNA vaccines against Covid-19.
“To be innovative and not redo what other people did.”
With your experience in academia and starting a company, what advice would you give to young researchers?
To be innovative and not redo what other people did. Also, to not stick too much to the textbook knowledge, of which not all is written in stone. As it was the case of mRNA, which was described as too fragile to be used for therapeutic applications. Dare to go a different route than everybody else, which is not without risk.
That said, I am not rich, I was two years without a job, I am not a professor and have a 20 square meter lab, so I am not sure if I am the right person to give career advice, since a lot of people my age are doing much better than me. Still, even if the price to pay was a slower career and lower salary, I am still happy with the career path I took.
Biography Steve Pascolo
Steve Pascolo studied Biology & Biochemistry at the Ecole Normale Supérieure in Paris, France and then conducted his PhD studies as well in Paris at the Institute Pasteur. In 1998, he moved to Tübingen, Germany for his postdoc in the lab of Hans-Georg Rammensee. In 2000, he co-founded Curevac and served as its Chief Scientific Officer until 2006. Afterwards he moved to the University Hospital Zurich, where is he currently a private lecturer and group leader in the Dermatology Department and head of the mRNA platform of the University of Zurich’s University Research Priority Program Cancer. In 2008, he founded Miescher Pharma, which provides consulting services and licenses a patented invention on Protamine mRNA. In 2017, he became an associate member of the NCCR RNA & Disease.
Interview by Dominik Theler conducted on April 26, 2021.