JAZF1 tips the balance in metabolic stress responses
A tight control of blood glucose levels is essential to ensure a normal body function. The pancreatic B-cells are key players by secreting the hormone insulin, which mediates the cellular glucose uptake and thus lowers the blood sugar levels. Pancreatic B-cell function and the insulin secretory pathway underly an intricate transcriptional network that allows fast and coordinated responses, which are required upon changes in glucose (i.e. after a meal) or cytokine concentrations (i.e. following infection). Disturbances in the insulin secretory pathway can manifest in metabolic disorders, such as type 2 diabetes (T2D). T2D accounts for 90% of the 420 million diabetic patients worldwide and represents a complex metabolic disease ultimately causing life-threatening health complications. Thus, it is of importance to understand the mechanism underlying the transcriptional network interactions causing the disease to develop novel therapeutic strategies.
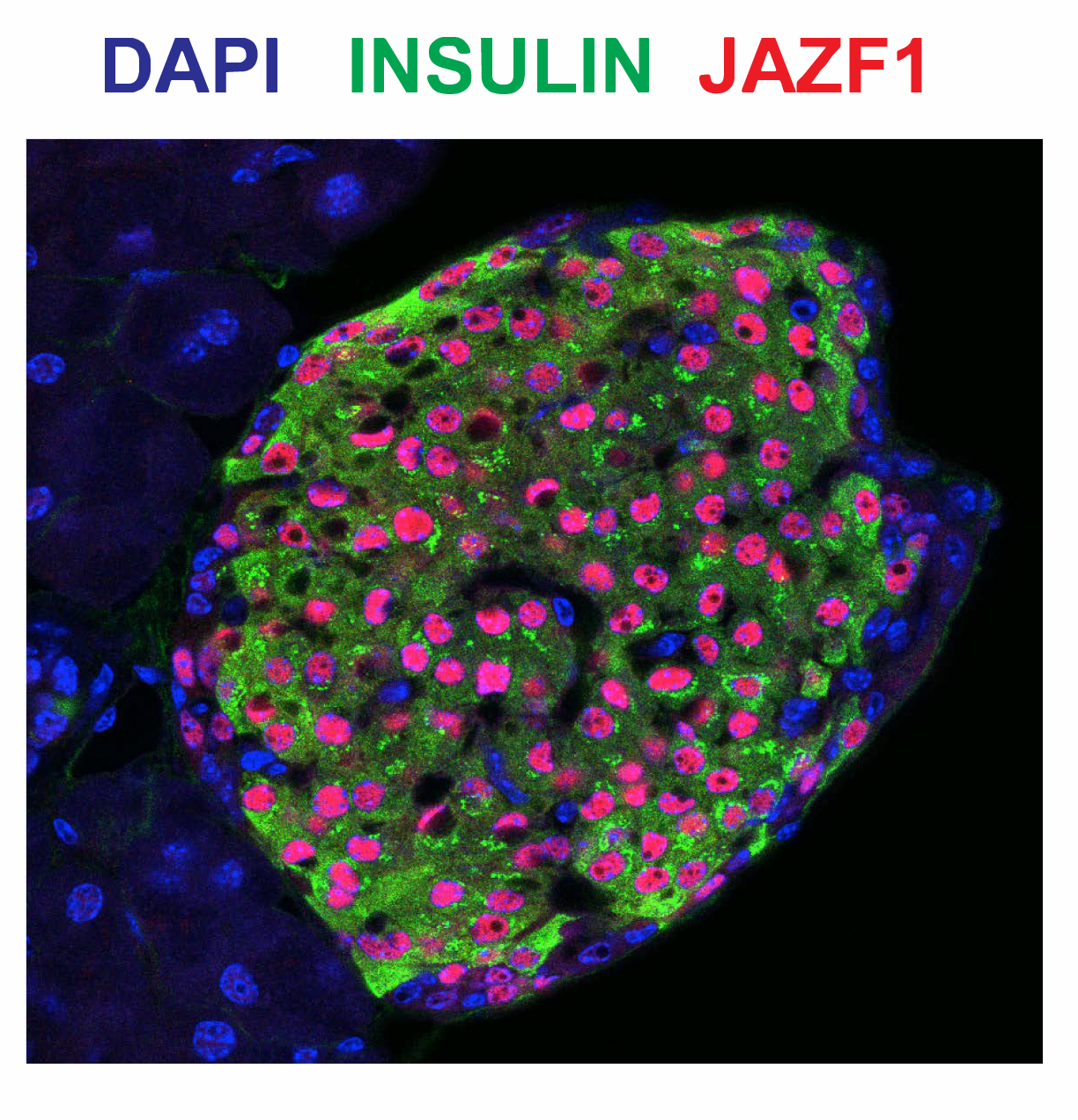
In a recent publication in Cell Reports, a team from the Stoffel lab at ETH Zurich in collaboration with endocrinologists and pathologists from the University Hospital Zurich investigated the cellular functions of JAZF1, a gene previously associated with T2D susceptibility. Initial characterization of JAZF1 expression revealed that JAZF1 expression is lowered in T2D patients and it re-localizes from cytoplasm to nucleus upon stimulation with glucose in pancreatic B-cells.
These results encouraged the authors to further seek for a better understanding of the cellular function of JAZF1 in metabolic stress conditions. By analyzing the functional consequences of JAZF1 loss in mouse and cellular models, they found that JAZF1 deficiency predisposes to ER stress, activation of the p53 pathway and ultimately results in apoptosis during metabolic stress conditions. This is accompanied by a reduction of pancreatic B-cell mass, a phenotypic hallmark that can often be found in T2D patients.
But what is the molecular explanation of the ER stress/apoptosis susceptibility of Jazf1-deficient cells? Kobiita and colleagues performed various experiments on the genomic and transcriptomic scale to identify a set of target genes regulated by JAZF1, which were further characterized in follow-up assays. These experiments revealed a multilayered transcriptional regulation of JAZF1 that ensures the fidelity of translation upon metabolic stress conditions: JAZF1 is a transcriptional regulator of ribosome biogenesis and translation, regulates the expression of aminoacyl-tRNA synthetases, and directly negatively regulates the expression of insulin. “It is challenging, but also fascinating that this factor is a positive regulator for many genes, but a negative regulator for others”, Markus Stoffel, corresponding author of the study, states.
Concluding a wealth of generated data, the researchers propose a model for the function of JAZF1: Under normal non-glycemic conditions, JAZF1 is dispensable: It is localized to the cytoplasm and is lingering until environmental conditions change. Upon elevated glucose levels, the cell can rapidly react to the stimulus by re-localizing JAZF1. In the nucleus, JAZF1 acts as a transcriptional regulator and ensures a cellular environment that allows to cope with the increased demand for insulin production: JAZF1 controls the fidelity of ribosome biogenesis and translation to prevent ER stress and subsequent cellular damage. Under chronic stress such as in diabetes, however, the overall levels of JAZF1 drop by a yet to determined mechanism. This relieves the JAZF1-mediated transcriptional regulation, ribosomal defects accumulate and translation goes out of balance. Resulting ER stress and the activation of the p53 pathway causes apoptosis and degradation of pancreatic B-cell mass. Markus Stoffel is excited about the therapeutic implications of the new findings: “Re-boosting the activity of JAZF1 in diabetic B-cells is a promising therapeutic approach that we will be following up in future.”
Kobiita et al. (2020) Cell Rep, 32(1), 107846
Text: Veronika Herzog