Keeping RNA helicases in check
RNA helicases are a highly conserved class of proteins that play central roles in regulatory processes and are fundamental in nearly every aspect of RNA metabolism. On the molecular level, RNA helicases bind and remodel RNA-protein complexes in an ATP-dependent manner. The DEAH RNA helicases, an essential family of RNA helicases, have pivotal roles in pre-mRNA splicing and ribosome biogenesis. Like many helicases, they harbour low intrinsic activity and no target specificity, and thus, they require dedicated adaptor proteins to fulfil their specific functions and get simultaneously activated. G-patch proteins are one broad class of such DEAH adapters and activators. With their eponymous glycine-rich motif these proteins hook the helicases to a target RNP and trigger RNA unwinding. The molecular mechanism of how G-patch proteins activate helicases, however, remained enigmatic.
In a recent study published in the Proceedings of the National Academy of Sciences, the Jonas Group at ETH Zurich investigated the structural basis of how the human DEAH helicase DHX15 is activated by the G-patch protein NKRF, which is involved in human ribosome biogenesis. To dissect the molecular mechanism of how the G-patch motif stimulates the helicase activity, the authors solved the crystal structure of DHX15 in complex with the G-patch motif of NKRF and followed up the structural analyses with rigorous biochemical assays.
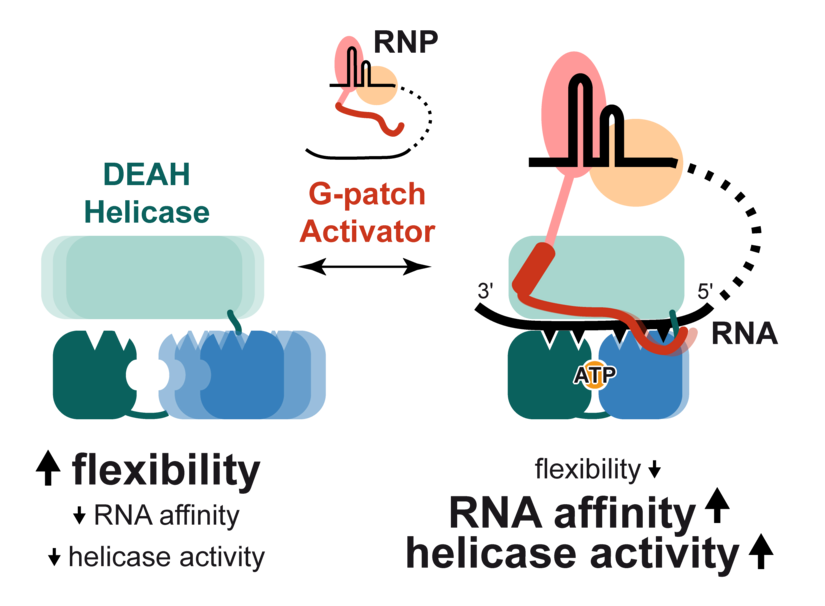
The crystal structure revealed how the G-patch peptide binds to DHX15: The mostly unstructured G-patch motif stretches along the DHX15 surface, almost like a molecular clamp that holds together two parts of the helicase that are otherwise highly flexible. Stefanie Jonas explains that the crystal structure was the key result of the project: “It was one of the rare events, where the structure already hinted strongly towards a molecular mechanism that we were able to confirm in a series of in vitro assays”.
The authors compared the structure with published structures from a fungal DHX15 ortholog that were solved in presence and absence of RNA. This analysis revealed that the conformation of the human DHX15 protein in presence of the G-patch peptide harbours an intact RNA channel, proposing that this conformation has a high affinity for RNA. Furthermore, a comparison of the solved DHX15-G-patch complex structures in presence or absence of ATP suggested that the tightened conformation of DHX15 still permits motions in the catalytic core. The researchers confirmed these observations in in vitro experiments: The restricted conformation of DHX15 induced by the G-patch peptide greatly enhances RNA affinity, ATPase and helicase activity.
Taken together, Studer et al. propose a new model of how DEAH RNA helicases are stimulated by the G-patch motif. In the absence of G-patch, DHX15 is highly flexible, has low RNA affinity and low ATPase activity. Upon G-patch binding, the helicase is restricted in its flexibility, which enhances RNA binding and catalytic activity DHX15. Thus, the specific activation of DHX15 by an adaptor protein restricts unproductive helicase movements on inadvertent targets in the cell. This detailed dissection of the mode of DHX15 activation sheds new light into how RNA helicases can fulfil their dedicated cellular functions. The mechanism of G-patch mediated DHX15-activation seems to be a general principle of helicase activation. Stefanie Jonas is excited about the broader significance of this study: “By taking our data and reanalysing the published structures of other helicases, we can draw conclusions beyond the DHX15 helicase and predict the mode of activation for a multitude of other RNA helicases.”
Studer et al. (2020) PNAS, 117(13):7159-7170 (Open Access)
Text: Veronika Herzog