The Integrator complex represses transcription of mRNAs and processes 3'-ends of non-coding RNAs. A new paper from the Jonas lab identifies a nucleic acid binding module of Integrator that brings RNA targets and nuclease subunit into proximity. The paper involved a collaboration with the NCCR RNA & Disease Structural MS Platform.
Abstract
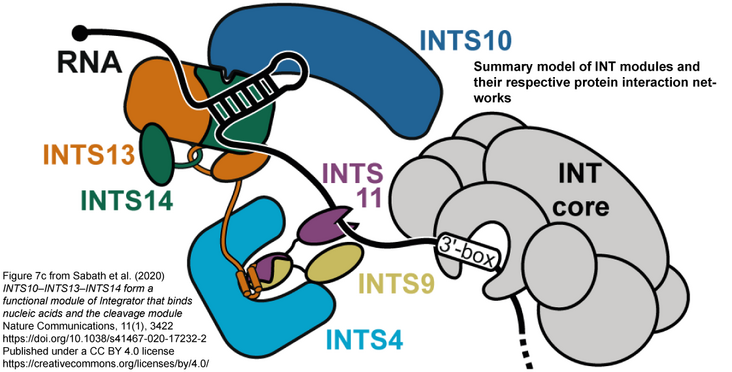
The Integrator complex processes 3'-ends of spliceosomal small nuclear RNAs (snRNAs). Furthermore, it regulates transcription of protein coding genes by terminating transcription after unstable pausing. The molecular basis for Integrator's functions remains obscure. Here, we show that INTS10, Asunder/INTS13 and INTS14 form a separable, functional Integrator module. The structure of INTS13-INTS14 reveals a strongly entwined complex with a unique chain interlink. Unexpected structural homology to the Ku70-Ku80 DNA repair complex suggests nucleic acid affinity. Indeed, the module displays affinity for DNA and RNA but prefers RNA hairpins. While the module plays an accessory role in snRNA maturation, it has a stronger influence on transcription termination after pausing. Asunder/INTS13 directly binds Integrator's cleavage module via a conserved C-terminal motif that is involved in snRNA processing and required for spermatogenesis. Collectively, our data establish INTS10-INTS13-INTS14 as a nucleic acid-binding module and suggest that it brings cleavage module and target transcripts into proximity.
Read the Publication in Nature Communications (Open Access)
Website NCCR Structural MS Platform
Abstract and figure from Sabath K. et al. (2020) Nature Communications, 11(1):3422 published under the CC BY 4.0 License.